Meet the JCMG Podiatry Doctors
Welcome to JCMG Podiatry, Mid-Missouri specialists in foot and ankle care. Our podiatrists strive to provide patients with evidence based medical and surgical options. If you are looking for a foot and ankle surgeon in Jefferson City, Cole County, Boone County or areas beyond, contact us today. We routinely accept patients from out of state and our staff is here to assist you with arranging telehealth consultations and to answer any of your questions. We look forward to addressing your needs.
Conditions We Treat
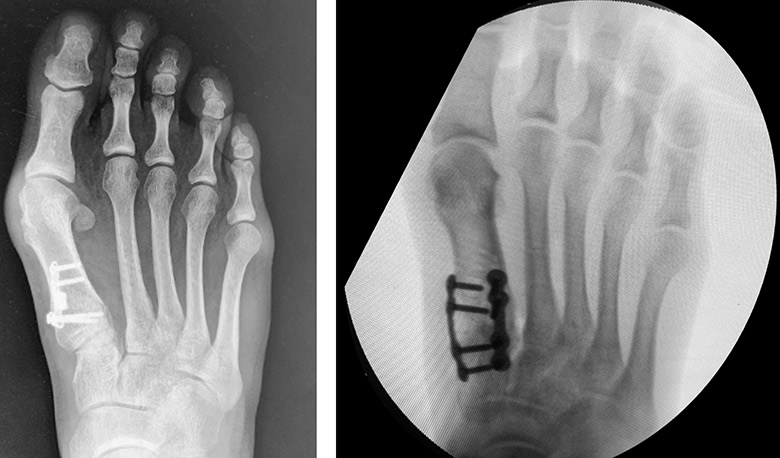
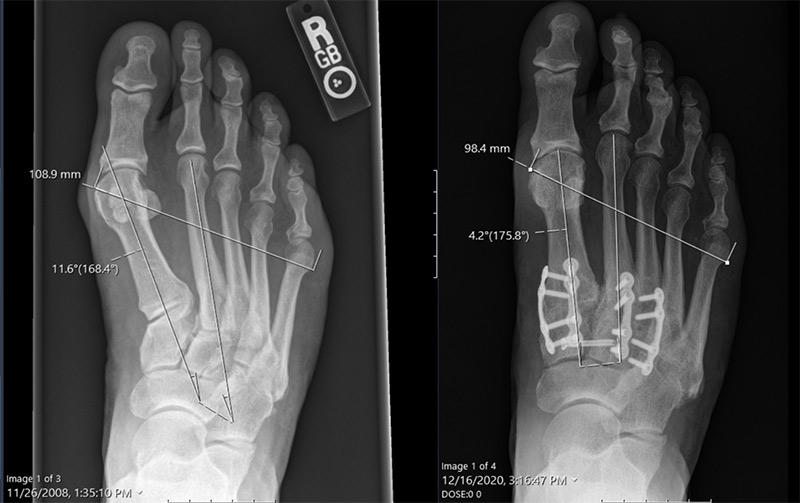
3D Lapiplasty Bunion Correction
Lapiplasty is a state-of-the-art procedure for correcting bunions. The benefits of Lapiplasty include:
- Long-lasting, permanent correction
- Corrects the deformity at the apex
- Quickly return to exercise and sports
- Covered by most insurance plans
- Can be used to correct recurrent deformities that have previously undergone failed 2D traditional surgery
Lapiplasty Procedures:
Featured Videos
Dr. McAleer Interviewed on The DoctorsDr. McAleer discusses Lapiplasty 3D Bunion Correction and real Lapiplasty patient, April, shares how it helped her get back to running marathons.
Lapiplasty 3D Bunion Correction: April’s StoryLearn about this procedure from April, a Lapiplasty patient, as well as Dr. J.P. McAleer of JCMG Podiatry.
Lapiplasty 3D Bunion Correction: Laura’s StoryLearn about this procedure from Laura, a Lapiplasty patient, as well as Dr. J.P. McAleer of JCMG Podiatry.
What is Lapiplasty 3D Bunion Correction?JCMG Podiatry doctors J.P. McAleer and William Duke explain how Lapiplasty works and what makes it such a groundbreaking technique.
What Our Patients Say
It was clear I had made the right choice to bring my three year old daughter to JCMG’s podiatrist Dr. Jody McAleer! I was just as scared as she was when she had jumped off the back of the couch and not a clue what to do next with the sudden pain! With excellent staff and quick care, Dr McAleer made our unforeseen experience a most pleasant one.
A couple of years ago I broke my fibula which ended up requiring surgical repair. I was recommended to seek treatment at JCMG Podiatry. I scheduled an appointment with Dr. McAleer and was immediately impressed by the friendliness and care from him and his staff. They were very attentive and informative of all procedures that would take place.
I am very pleased with the care my family has received and would highly recommend JCMG Podiatry. Both Dr. Duke and Dr. McAleer are very skilled but are also very approachable and spend time discussing the patient’s concerns with them. In addition, the staff at JCMG Podiatry is so friendly and supportive.
When I found out I had a fungus under my toenail I was devastated. I was too ashamed to go to the doctor, so I bought over-the-counter medication. It didn’t help. I finally went to see Dr. Duke. He prescribed me some medicine for my toenails. I received treatments with the laser and had amazing results. Thank you Dr. Duke!
I finally took the advice of my family and had my ankle examined by Dr. McAleer. An MRI revealed the damage I had done over my lifetime. Dr. McAleer helped guide me through my treatment options. He was able to repair my tendon and stabilize my ankle. I feel like myself again and I am more active than ever. Thanks to the doctors and staff of JCMG Podiatry.
Dr. Duke is a compassionate, honest and intelligent physician, and one that I trust my children with completely. His professional bedside manner is impressive, but his ability to treat my kids like his own and make them feel super comfortable in an office setting is something hard to come by.
The care provided by Dr. Duke and his entire staff was outstanding. Dr. Duke treated my kids as if they were his own, he provided excellent follow up care, and always gave my kids all the time they needed to answer their questions. I highly recommend Dr. Duke for any of your foot or ankle care needs.
Dr. Duke has treated my kids on several occasions for various reasons. Each time he was very knowledgeable and professional. He also has the ability to calm my kids when they are nervous about a procedure he is performing. He is honest, trustworthy, and truly enjoys helping people. I would highly recommend Dr. Duke!
Dr. McAleer had been treating me for a fungus under my toenails. During one of my follow-up visits I mentioned to him a pain I had been having around one of the metatarsals of my left foot. He was able to inject the area with steroids and my foot has been pain free since. From here on out, I will bring all of my foot problems to his attention. Thanks Doc!